THIS WEBSITE HAS MOVED TO "www.theamateurchemist.com". Please feel free to visit... :)
General Lab Equipment
In this section I will have a picture of some common pieces of glassware that you may have in your lab, along with a short description and name of the piece. Most descriptions will be taken from wikipedia.
Beaker
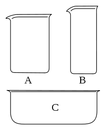
The common griffin beaker. A beaker is a simple container for stirring, mixing and heating liquids commonly used in many laboratories, first designed in 1453. Beakers are generally cylindrical in shape, with a flat bottom. Most also have a small spout (or "beak") to aid pouring as shown in the picture. Beakers are available in a wide range of sizes, from one millilitre up to several litres.
(A) A low-form or Griffin form beaker (B) A tall-form or Berzelius beaker (C) A flat beaker or crystallizer
(A) A low-form or Griffin form beaker (B) A tall-form or Berzelius beaker (C) A flat beaker or crystallizer
Erlenmeyer Flask

The common Erlenmeyer flask. An Erlenmeyer flask, also known as a conical flask, is a widely used type of laboratory flask which features a flat bottom, a conical body, and a cylindrical neck. It is named after the German chemist Emil Erlenmeyer, who created it in 1861. It is almost always used in titration and for heating liquids. Many times, it is preferred over a regular beaker because you can put a stopper in it.
Round Bottom Boiling Flask

Round-bottom flasks are types of flasks having spherical bottoms used as laboratory glassware, mostly for chemical or biochemical work. They are typically made of glass for chemical inertness; and in modern days, they are usually made of heat-resistant borosilicate glass. There is at least one tubular section known as the neck with an opening at the tip. Two or three-necked flasks are common as well. Round bottom flasks come in many sizes, from 5 mL to 20 L, with the sizes usually inscribed on the glass. The ends of the necks are usually conical (female) ground glass joints. These are standardized, and can accept any similarly-sized tapered (male) fittings. Standard Taper 24/40 is common for 250 mL or larger flasks, while smaller sizes such as 14 or 19 are used for smaller flasks.
Because of the round bottom, cork rings are needed to keep the round bottom flasks upright. When in use, round-bottom flasks are commonly held at the neck by clamps on a stand. The round bottoms on these types of flasks allow more uniform heating and/or boiling of liquid. Thus, round-bottom flasks are used in a variety of applications where the contents are heated or boiled. Round-bottom flasks are usually used in distillation by chemists as distilling flasks and receiving flasks for the distillate. This flask shape is also more resistant to fracturing under vacuum, as a sphere more evenly distributes stress across its surface.
Round-bottom flasks are often used to contain chemical reactions run by chemists, especially for reflux set-ups and laboratory-scale synthesis. Boiling chips are often added in distilling flasks for distillations or boiling chemical reactions to allow a nucleation site for gradual boiling. This nucleation avoids a sudden boiling surge where the contents may overflow from the boiling flask. Stirring bars or other stirring devices suited for round-bottom flasks are sometimes used. Round bottom flasks suffer from poor stirring when compared with Erlenmeyer flasks, as they can't accept large stir bars and material can become trapped at the base. For a reflux set-up, a condenser is typically attached to the middle or only neck of the flask being used. Additional necks on a flask could allow a thermometer or a mechanical stirrer to be inserted into the flask contents. The additional necks can also allow a dropping funnel to be attached to let reactants slowly drip in.
Special electrically powered heating mantles are available in various sizes into which the bottoms of round-bottom flasks can fit so that the contents of a flask can be heated for distillation, chemical reactions, boiling, etc. Heating can also be accomplished by submerging the bottom of the flask into a heat bath, water bath, or sand bath. Similarly cooling can be accomplished by partial submerging into a cooling bath, filled with e.g. cold water, ice, eutectic ice/salt mixtures, dry ice/solvent mixtures, or liquid nitrogen.
Liebig Condenser

In a laboratory a condenser is a piece of laboratory glassware used to cool hot vapors or liquids. A condenser usually consists of a large glass tube containing a smaller glass tube running its entire length, within which the hot fluids pass.
The ends of the inner glass tube are usually fitted with ground glass joints which are easily fitted with other glassware. During reflux, the upper end is usually left open to the atmosphere or vented through a bubbler or a drying tube to prevent the ingress of water or oxygen. The outer glass tube usually has two hose connections, and a coolant (usually tap water or chilled water/anti-freeze mixture) is passed through it. For maximum efficiency, and to maintain a smooth and correctly directed thermal gradient so as to minimize the risk of thermal shock to adjacent glassware, the coolant usually enters through the lower fitting, and exits through the higher fitting. The liebig condenser is the most simple type of condenser. It is simply an inner tube through which the vapors pass through and an outer tube through which the coolant passes through. If your tight on money, get this type of condenser, as it can be used for all kind of distillations, and for reflux. There are many other types of condensers. Below, you can see how a fractional distillation works.
Separatory Funnel
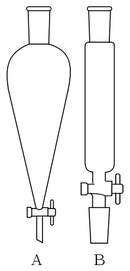
A separatory funnel, is a piece of laboratory glassware used in liquid-liquid extractions to separate (partition) the components of a mixture into two immiscible solvent phases of different densities. Typically, one of the phases will be aqueous, and the other a non-polar lipophilic organic solvent such as ether, MTBE, dichloromethane, chloroform, or ethyl acetate. All of these solvents form a clear delineation between the two liquids. The two layers formed are usually known as the organic and aqueous phases. Most organic solvents float on top of an aqueous phase, though important exceptions are most halogenated solvents. The organic solvent used for the extraction must not react with the substance to be extracted or with water. It should also have a low boiling point so it can be easily removed from the product.
A separating funnel takes the shape of a cone with a hemispherical end. It has a stopper at the top and stopcock (tap), at the bottom. Separating funnels used in laboratories are typically made from borosilicate glass and their stopcocks are made from glass or PTFE. Typical sizes are between 50 mL and 3 L.

