THIS WEBSITE HAS MOVED TO "www.theamateurchemist.com". Please feel free to visit... :)
How to make nitrocellulose (guncotton)
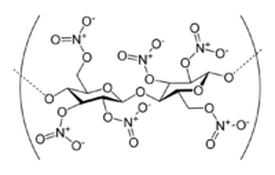
Nitrocellulose (also: cellulose nitrate, flash paper, flash cotton, flash string) is a highly flammable compound formed by nitrating cellulose through exposure to nitric acid or another powerful nitrating agent. When used as a propellant or low-order explosive, it was originally known as guncotton (Wikipedia).
Making nitrocellulose is quite easy. All you need is some nitric acid which has at least a concentration of 70% and some concentrated 98% sulfuric acid. It's a pretty cool demonstration as well. Just tell people you are going to take this piece of cotton, put it on your hand and light it on fire. Then you can see their reaction when it goes up in a giant fireball and your hand isn't even burnt.
Ingredients
70ml 98% H2SO4
30ml 70% HNO3
5 grams cotton
Procedure
First, add the nitric acid to the sulfuric acid slowly. You can use an ice bath if you want to help prevent decomposition of the nitric acid into nitrogen oxides. I have a pretty good fume hood, so I don't usually do this. Then, add the cotton to this mixture. It will probably absorb most of the liquid. Make sure all of the cotton is fully wet, so it all reacts. Let this react for around 30-45 minutes. The exact time doesn't matter much.
Now, you must fully get rid of the acid from the nitrocellulose. I usually take the cotton out of the acid, wash it in water, then wash in a different glass of water, then in some sodium bicarbonate, then water again, and then maybe some sodium bicarbonate again. Just make sure all of the acid is out. You can then dry it. It helps if you pull the cotton apart a bit, plus it will burn better.
When I first made it, my nitric acid was not strong enough, around 40-50% I think, and it didn't nitrate fully. In fact, the cotton just sort of fell apart. The next couple of times I did it worked very well. I had used white fuming nitric acid though, for some stupid reason, and it had dissolved the cap of the bottle it was stored in and became quite yellow in color. It worked well, my guncotton was just a bit yellow. I have heard you can also do this with a metal nitrate such as as potassium or sodium nitrate and sulfuric acid, but I could never get it to work right.
I am not liable for anything you do!!! Read the Disclaimer Section!
Making nitrocellulose is quite easy. All you need is some nitric acid which has at least a concentration of 70% and some concentrated 98% sulfuric acid. It's a pretty cool demonstration as well. Just tell people you are going to take this piece of cotton, put it on your hand and light it on fire. Then you can see their reaction when it goes up in a giant fireball and your hand isn't even burnt.
Ingredients
70ml 98% H2SO4
30ml 70% HNO3
5 grams cotton
Procedure
First, add the nitric acid to the sulfuric acid slowly. You can use an ice bath if you want to help prevent decomposition of the nitric acid into nitrogen oxides. I have a pretty good fume hood, so I don't usually do this. Then, add the cotton to this mixture. It will probably absorb most of the liquid. Make sure all of the cotton is fully wet, so it all reacts. Let this react for around 30-45 minutes. The exact time doesn't matter much.
Now, you must fully get rid of the acid from the nitrocellulose. I usually take the cotton out of the acid, wash it in water, then wash in a different glass of water, then in some sodium bicarbonate, then water again, and then maybe some sodium bicarbonate again. Just make sure all of the acid is out. You can then dry it. It helps if you pull the cotton apart a bit, plus it will burn better.
When I first made it, my nitric acid was not strong enough, around 40-50% I think, and it didn't nitrate fully. In fact, the cotton just sort of fell apart. The next couple of times I did it worked very well. I had used white fuming nitric acid though, for some stupid reason, and it had dissolved the cap of the bottle it was stored in and became quite yellow in color. It worked well, my guncotton was just a bit yellow. I have heard you can also do this with a metal nitrate such as as potassium or sodium nitrate and sulfuric acid, but I could never get it to work right.
I am not liable for anything you do!!! Read the Disclaimer Section!