THIS WEBSITE HAS MOVED TO "www.theamateurchemist.com". Please feel free to visit... :)
Synthesis of Hydrobromic Acid
Hydrobromic acid is a strong acid formed by dissolving the diatomic molecule hydrogen bromide in water. It is used in may organic syntheses and is a very important chemical. It is a stronger acid then Hydrochloric, but still weaker than Hydroiodic acid.
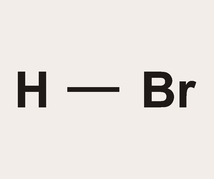
Formula: HBr
Density: 1.49 g/cm³
Boiling point: 251.6° F (122° C)
Melting point: 12.2° F (-11° C)
Soluble in: Water
2NaBr + H2SO4 ---> Na2SO4 + 2HBr
Ingredients: 240 grams sodium/potassium bromide
400 ml water
180 ml 98% sulfuric acid
Procedure: First, dissolve the sodium bromide in water using a magnetic stirrer. Make sure it is all dissolved. You can use potassium bromide as well. Next, place the beaker in an ice bath, and slowly add the sulfuric acid. Never let the temperature rise over 75 degrees Celsius, otherwise some bromine may be formed.
Let the beaker warm to room temperature, and filter the precipitated sodium bisulfate out. Use multiple filter papers, as the acid can eat through them if they are not strong enough.
The filtrate is distilled from a 1 liter flask. The water comes over 1st. Collect the fraction that comes over between 124 and 127 degrees Celsius. To further purify, distill again after adding a little bit of barium bromide to the flask. The final yield should be somewhere around 285 grams, which is 85% of the theoretical yield.
I will post some pictures as soon as I attempt this myself.
I am not liable for anything you do!!! Read the Disclaimer Section!
Formula: HBr
Density: 1.49 g/cm³
Boiling point: 251.6° F (122° C)
Melting point: 12.2° F (-11° C)
Soluble in: Water
2NaBr + H2SO4 ---> Na2SO4 + 2HBr
Ingredients: 240 grams sodium/potassium bromide
400 ml water
180 ml 98% sulfuric acid
Procedure: First, dissolve the sodium bromide in water using a magnetic stirrer. Make sure it is all dissolved. You can use potassium bromide as well. Next, place the beaker in an ice bath, and slowly add the sulfuric acid. Never let the temperature rise over 75 degrees Celsius, otherwise some bromine may be formed.
Let the beaker warm to room temperature, and filter the precipitated sodium bisulfate out. Use multiple filter papers, as the acid can eat through them if they are not strong enough.
The filtrate is distilled from a 1 liter flask. The water comes over 1st. Collect the fraction that comes over between 124 and 127 degrees Celsius. To further purify, distill again after adding a little bit of barium bromide to the flask. The final yield should be somewhere around 285 grams, which is 85% of the theoretical yield.
I will post some pictures as soon as I attempt this myself.
I am not liable for anything you do!!! Read the Disclaimer Section!